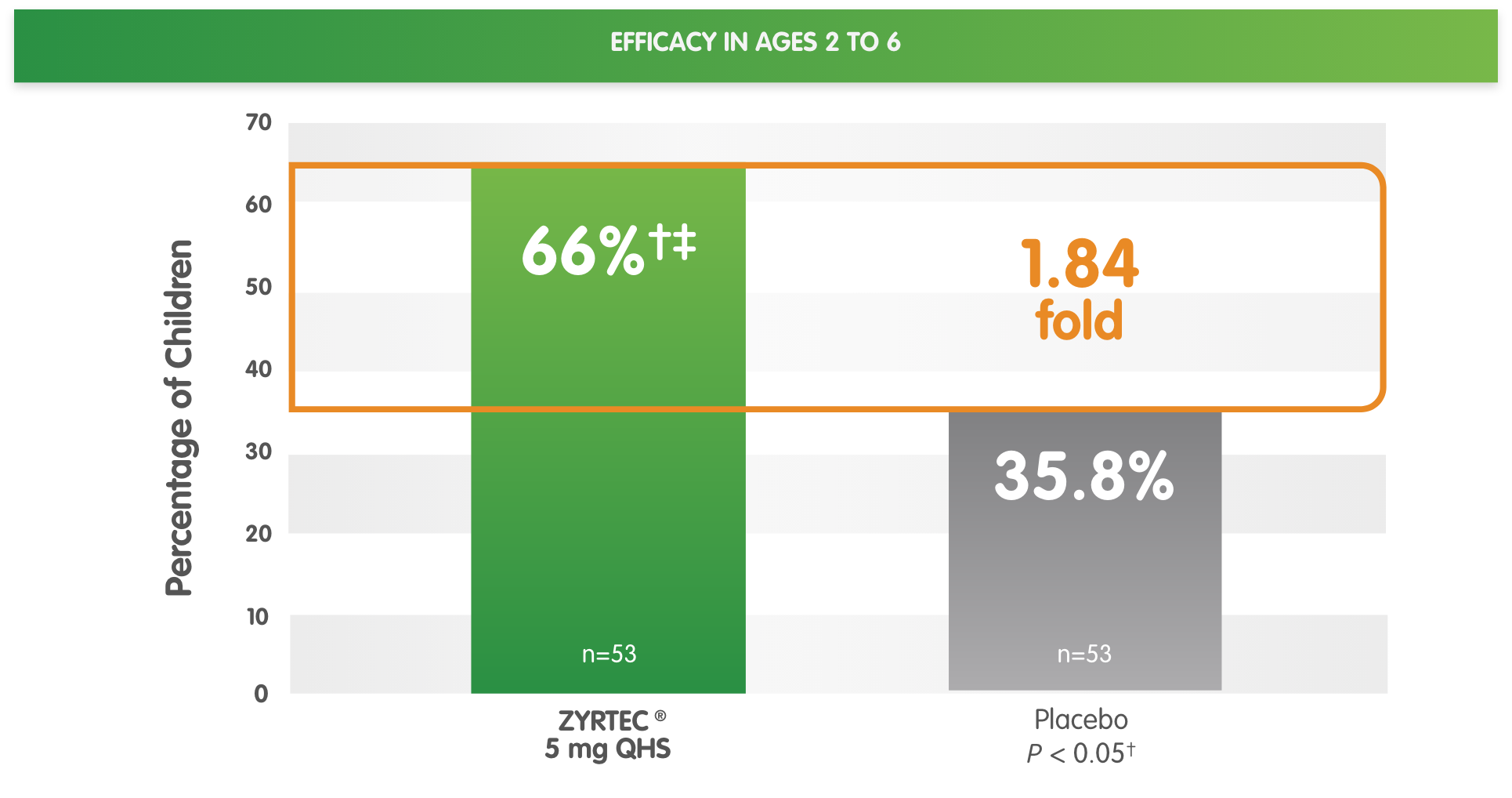
*Randomized, placebo-controlled study of children aged 2 to 6 years with SAR during a 2-week period. Primary endpoint was the change in total symptom score complex from baseline.
†Significant vs placebo.
‡ZYRTEC® 5 mg vs placebo ratio at ≥50% reduction is 1.84-fold.
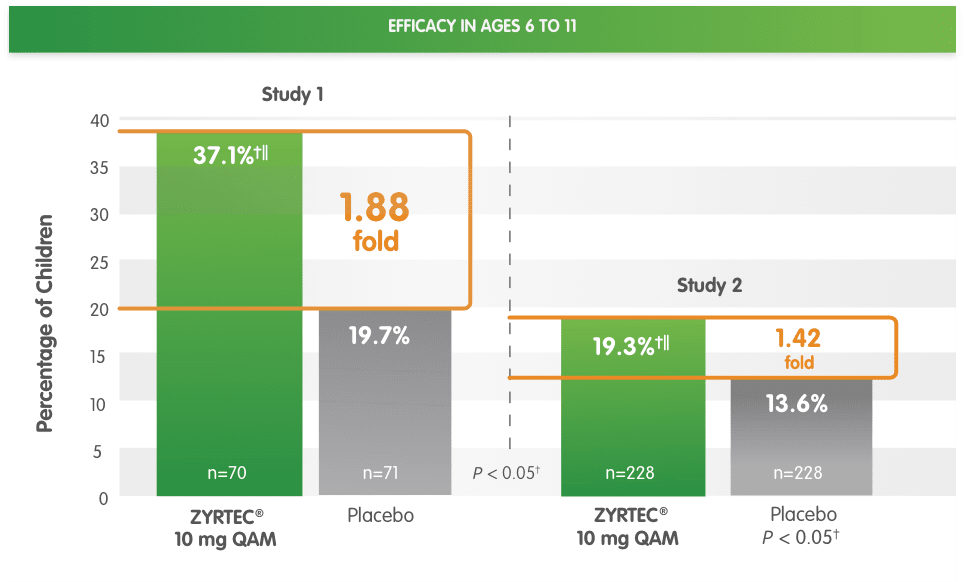
§Randomized, placebo-controlled studies of children aged 6 to 11 years with SAR during a 2-week period. Primary endpoint was the change in total symptom score complex from baseline.
||ZYRTEC® 10 mg vs placebo ratio at ≥50% reduction is 1.88-fold in Study 1 and 1.42-fold in Study 2.
References:
1. Data on file. Johnson & Johnson Consumer Inc., McNeil Consumer Healthcare Division.