Tolerability
ZYRTEC® is generally well tolerated by children1
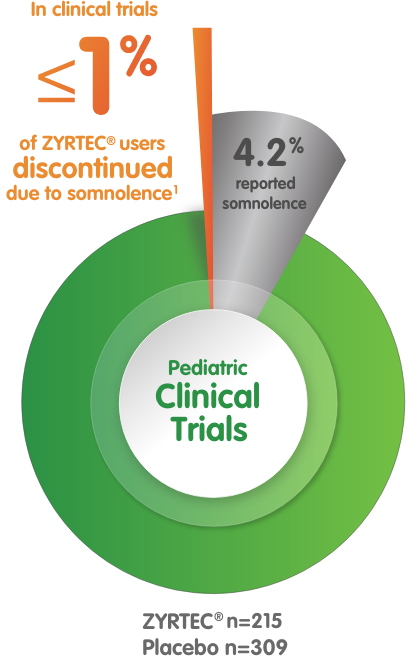
In studies of children ages 6 to 11 years:
- 4.2% of patients taking ZYRTEC® 10 mg reported somnolence (n=215) vs 1.3% with placebo (n=309)1
- ≤1% of ZYRTEC® patients discontinued due to somnolence1


A story for caregivers to share with kids
Popular 2-part set helps families manage allergies. Available to download in English and Spanish.


Free monthly Bundle Box supports your pediatric patient care
Samples and resources from Children’s ZYRTEC® and other Kenvue Pediatric products.
Reference: 1. Data on file. Johnson & Johnson Consumer Inc., McNeil Consumer Health Division.
