The ZYRTEC® Difference
Start with ZYRTEC® as your foundation for allergy relief
Use ZYRTEC® as your foundation for allergy relief, whether patients’ symptoms are mild, moderate, or severe.
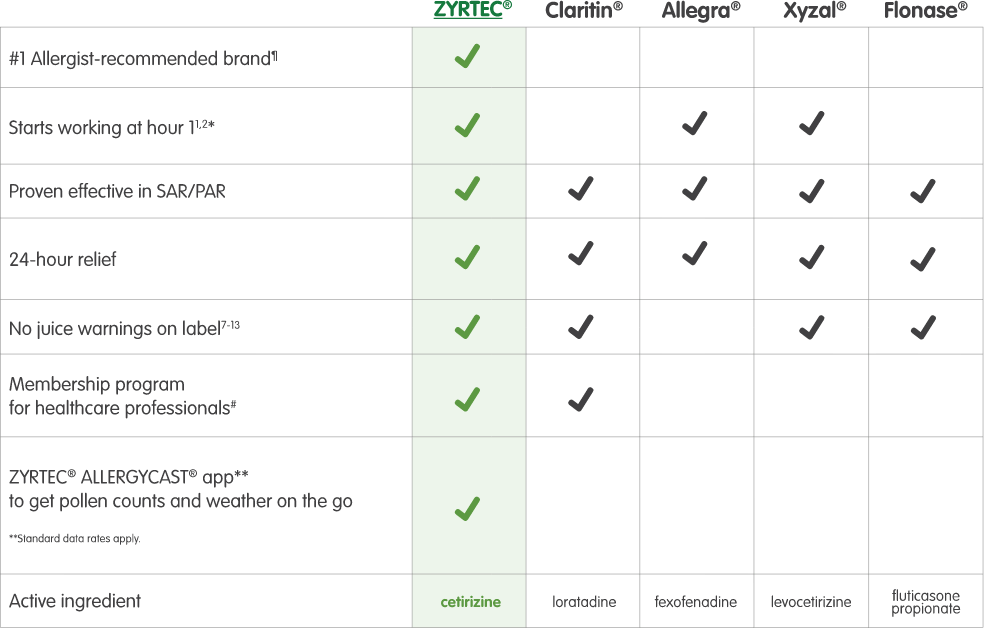
ZYRTEC® starts working at hour 1 the first day you take it; Claritin®, at hour 31,2*
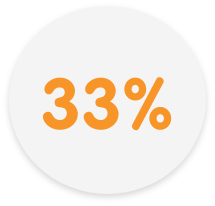
Delivered 33% greater relief than Allegra® at 21 to 24 hours3†
Relieves bothersome ocular symptoms such as itchy, watery eyes4
Significantly improved allergy-related quality of life in clinical trials5,6‡§||
Children’s ZYRTEC® offers convenient once-daily dosing in flavors kids will love

Has no juice warning on label, unlike Allegra®
See how ZYRTEC® compares to other 24-hour OTC allergy medications
ZYRTEC® contains cetirizine, an established molecule for effective allergy relief.
PAR = perennial allergic rhinitis; SAR = seasonal allergic rhinitis.

Allegra® has a juice warning. ZYRTEC® does not.
Fruit juice may reduce the bioavailability and effectiveness of
Allegra®.7,12,13 ZYRTEC® does not have a juice interaction statement on its label.9




Do you know the entire ZYRTEC® family of products?
Different formulations meet patients’ varied needs.
*ZYRTEC® 10 mg starts working at hour 1 and Claritin® starts working at hour 3, based on first dose on the first day of a 2-day study in 2 pollen-chamber studies. Primary endpoint measured mean improvement from baseline in Major Symptom Complex (MSC) severity score. MSC symptoms included runny nose, sniffles, itchy nose, nose blows, sneezes, and watery eyes.
†Based on first dose on the first day of a 2-day pollen-chamber study with ZYRTEC® 10 mg vs Allegra® 180 mg at hours 21 to 24. Primary efficacy endpoint was change in total symptom severity score from baseline at hours 21 to 24. Total symptom severity complex score was defined as the sum of self-assessed severity scores of 4 symptoms: runny nose, sneezing, itchy nose/palate/throat, and itchy/watery eyes.
‡Randomized, placebo-controlled, 2-week study of adult patients with seasonal allergic rhinitis (N=431 for each treatment group).
§Impact on self-reported work/school-related productivity and activity impairment was assessed using the Work Productivity and Activity Impairment-Allergy Specific (WPAI-AS) questionnaire, which measures performance based on impairment at work (limitations in the amount or kind of work done, work accomplished, or work done as carefully as usual) and activity impairment (limitations in usual activities, such as work around the house, shopping, child care, exercising, etc).
||Patient disease-specific quality of life was assessed using the Rhinoconjunctivitis Quality of Life Questionnaire (RQLQ), which measures 7 domains: activity limitation, sleep problems, nose symptoms, eye symptoms, non–hay fever symptoms, practical problems, and emotional function.
¶Among OTC oral antihistamines.
#ZYRTEC® membership program for healthcare professionals is ZYRTEC ZIPLINE®.
References: 1. Day JH, Briscoe M, Widlitz MD. Cetirizine, loratadine, or placebo in subjects with seasonal allergic rhinitis: effects after controlled ragweed pollen challenge in an environmental exposure unit. J Allergy Clin Immunol. 1998;101(5):638-645. 2. Day JH, Briscoe M, Rafeiro E, Chapman D, Kramer B. Comparative onset of action and symptom relief with cetirizine, loratadine, or placebo in an environmental exposure unit in subjects with seasonal allergic rhinitis: confirmation of a test system. Ann Allergy Asthma Immunol. 2001;87(6):474-481. 3. Day JH, Briscoe MP, Rafiero E, Hewlett D, Chapman D, Kramer B. Randomized double-blind comparison of cetirizine and fexofenadine after pollen challenge in the environmental exposure unit: duration of effect in subjects with seasonal allergic rhinitis. Allergy Asthma Proc. 2004;25(1):59-68. 4. Patel M, Urdaneta E, Franklin K, et al. Cetirizine significantly relieves ocular allergy symptoms in subjects with seasonal allergic rhinitis. Poster Abstract. Data on file. Johnson & Johnson Consumer Inc., McNeil Consumer Health Division. 5. Murray JJ, Nathan RA, Bronsky EA, Olufade AO, Chapman D, Kramer B. Comprehensive evaluation of cetirizine in the management of seasonal allergic rhinitis: impact on symptoms, quality of life, productivity, and activity impairment. Allergy Asthma Proc. 2002;23(6):391-398. 6. Noonan MJ, Raphael GD, Nayak A, et al. The health-related quality of life effects of once-daily cetirizine HCI in patients with seasonal allergic rhinitis: a randomized double-blind, placebo-controlled trial. Clin Exp Allergy. 2003;3(3):351-358. 7. Allegra® Allergy 24 Hour Drug Facts. 8. Claritin® 24-Hour Drug Facts. 9. ZYRTEC® 24-Hour Drug Facts. 10. XYZAL® Allergy 24HR Drug Facts. 11. FLONASE® Drug Facts. 12. Allegra® [package insert]. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; 2007. 13. FAQs. Allegra®. Accessed May 20, 2021. https://www.allegra.com/en-us/faq-what-are-seasonal-allergies